HOW CLOVERS CURE ECOLI
Background: We are trying to find medicinal uses for plants by testing them for chemicals
Purpose: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials: Balance, weight boat, lab scoops, Inoculating loop, Ni/Cr wire, Syringe, LB broth base, Petri dishes, Media bottles, E. Coli
Procedure-
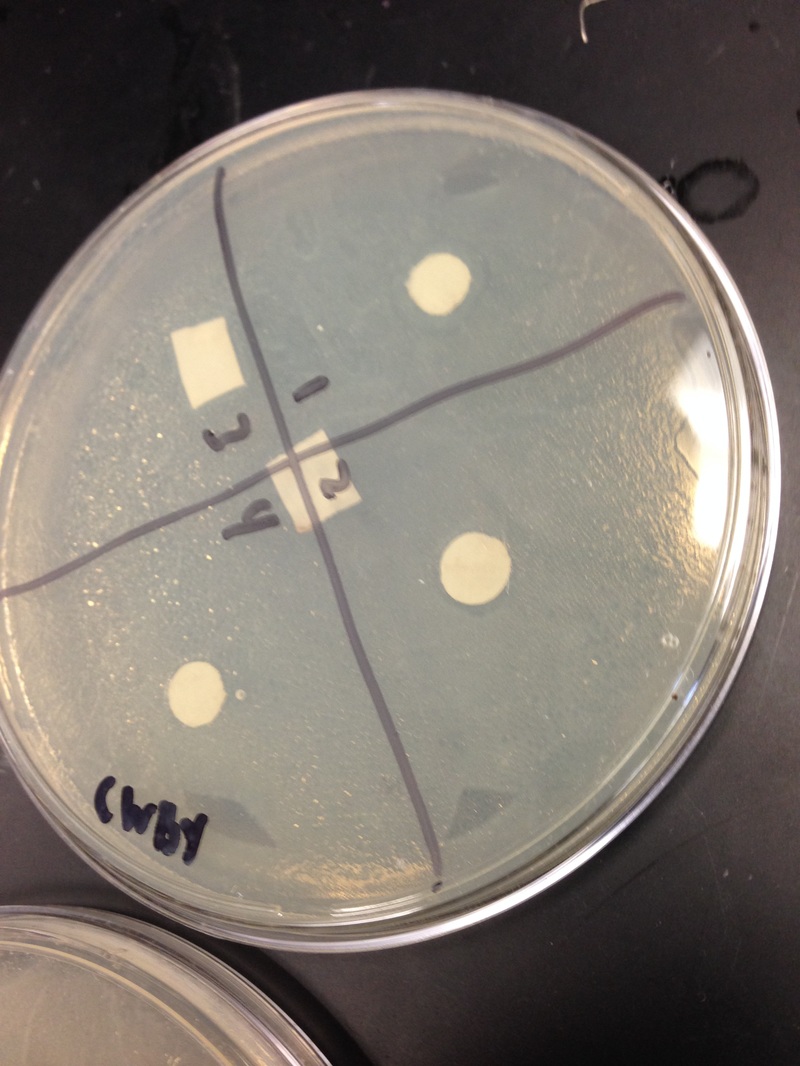
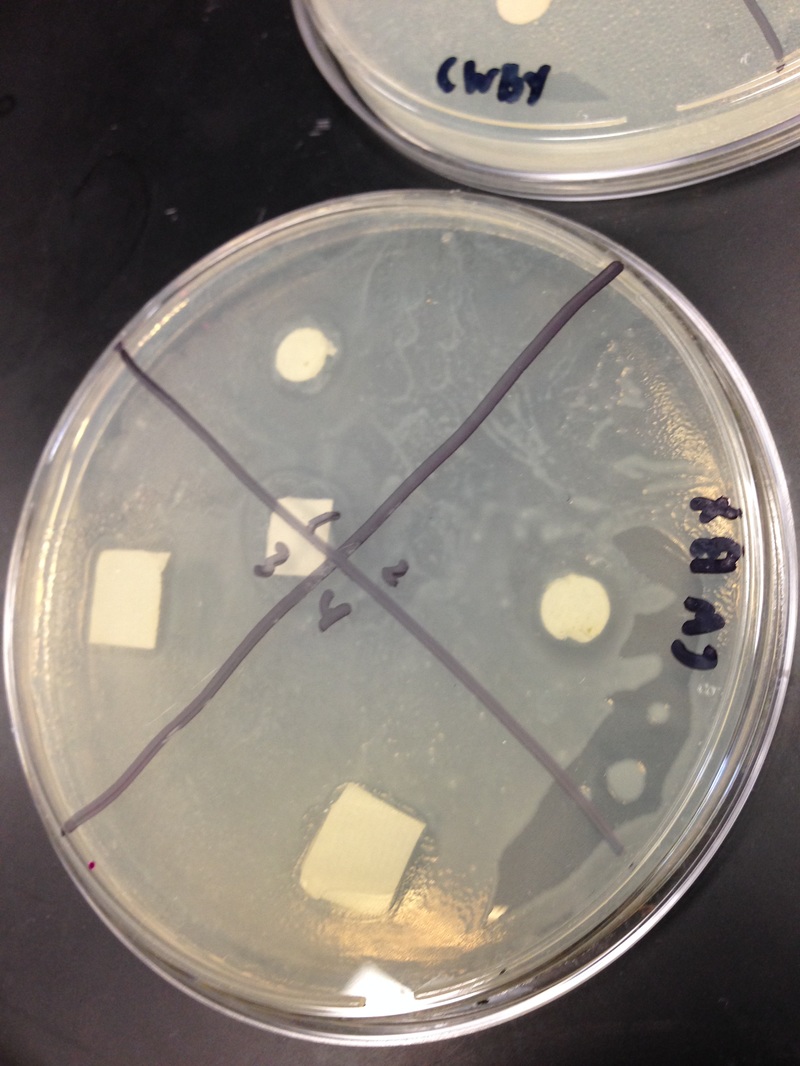
Results: Our results were as expected, on the first day, there was a bit of a ring around the paper. After that on the second day of incubation there was the clearance of the agar on the second day of incubation. In our water dish, the ampicillin did its job and there was a clearance. There was an obvious clearance of the agar. Our plant-water had a clearance as well and it was more obvious than the water. In our methanol, there was the same thing, on the first day there was a slight ring around the papers, but on the second day there was clearance ring around the papers. Overall the clearings around the filter disks for positive control and negative control was visible. The density of the bacterial lawn was not that dense but it was visible. There was no sign of contamination, and the petri dishes looked uncontaminated.
Purpose: What plant materials, found locally, contain active ingredients that will inhibit the growth of bacteria?
Materials: Balance, weight boat, lab scoops, Inoculating loop, Ni/Cr wire, Syringe, LB broth base, Petri dishes, Media bottles, E. Coli
Procedure-
- Preparing plant extracts:
- Use a mortar and paste to grind up 2 grams of plant tissue with 10 ml of deionized water
- Let it sit for 3 minutes
- Filter the sample through an 11 cm filter paper/funnel
- Filter/sterilize the extract using a syringe filter
- Collect 1 mL of the filter-standard extract into a 17 microtube. Label the sample
- Attach prefilter to syringe and rinse with water
- Take to Laminar hood: Plant extract, Syringe/prefilter, Microfuge tube rack, Pipet
- Label microfuge tube with initials and whether it is water/methanol and put in rack
- Attach the sterile filter to the prefilter
- Load 1.7 mL of extract to syringe using pipet
- Put plunger in and depress it
- Have at least 1 mL of sterilized extract
- Snap on cap on microfuge tube
- Evaporate methanol from methanol extracts by placing a tube, with a cap, upon a 65 degree heat-block overnight
- Reconstitute methanol extract 1 mL sterile deionized water
- Using sterile forceps place 3 sterile pieces of filter paper into the filtered extract 4 degree celsius
- Store until ready to use
- Draw a + on each plate bottom and number quadrants 1-4
- Liquify sterile LB agar in the microwave
- Using sterile technique pour approximately 20 mL of agar into Petri plate
- Using sterile forceps add the appropriate number of sterile disks to each tube of filtered extract
- Label both plates with either M for methanol or W for water
- Place the disks into the appropriate solution
- Plant extracts, syringe/ prefilter, pipet
- Label microfuge tube: Water and Methanol
- Attach sterile filter to pre filter
- Load 1.7 ml of extract into syringe, using pipet
- Depress plunger 1.0 ml filter sterilized extract
- Label agar plates with a cross, label quadrants
- Using sterile forceps, add the appropriate number of sterile disks to each tube of filtered extract
- Prepare negative control disks:
- 2 - sterile water
- 2- Ampicillin
- Place the disks in the appropriate solution
- Sterile disks were added to microfuge tubes containing one mL sterile water
- 10-20 mL of warmed nutrient agar was poured into 2 petri dishes using sterile technique
- After allowing agar to solidify, plates were turned upside down and stored at 4 degrees celsius overnight
- One mL of Ecoli colony was added to each plate. A flame-sterilized spreading loop was used to spread the bacteria throughout the surface of the agar
- Using flame-sterilized forceps, filter disks were placed in separate quadrants onto the plate in the following sequence: Water, plant extracts, ampicillin. Plants were left on lab bench for 20 min. to allow both bacteria and filter filter disks to adhere to the agar
- Plants were incubated upside down, overnight at 37 degrees celsius. Plate were photographed and observed for clearance around the filter disks after 24 hours, 48 hours, and 72 hours
Results: Our results were as expected, on the first day, there was a bit of a ring around the paper. After that on the second day of incubation there was the clearance of the agar on the second day of incubation. In our water dish, the ampicillin did its job and there was a clearance. There was an obvious clearance of the agar. Our plant-water had a clearance as well and it was more obvious than the water. In our methanol, there was the same thing, on the first day there was a slight ring around the papers, but on the second day there was clearance ring around the papers. Overall the clearings around the filter disks for positive control and negative control was visible. The density of the bacterial lawn was not that dense but it was visible. There was no sign of contamination, and the petri dishes looked uncontaminated.